


NYE IDEÉR (DOFIER)
milliarder kroner i samlet verdi
på
portefølje-selskaper
nye lisensavtaler
nye patentsøknader
nye kliniske studier
pågående kliniske studier
nye selskaper etablert
millioner kroner til ny forskning
millioner kroner innhentet
i privat kapital

FOTO: Festoon Lighting Brisbane
– hvordan ta forskning til markedet?
Inven2 sitt kjerneområde er å ta resultatene fra forskning til et produkt eller en tjeneste i et marked. Det gjøres gjennom en verdikjede som starter med idéspeiding og markedsføring av Inven2 sine tjenester rettet mot forskere ved både Universitetet i Oslo sykehusene i Helse Sør-Øst.
Les merInven2 arrangerer en rekke aktiviteter hvert år for å stimulere til innovasjon. Noe gjør vi alene, men mye gjør vi sammen med aktører i det store økosystemet vi er en del av.

for innovasjon
Samarbeid mellom næringsliv og forskning er viktig for mer innovasjon, og fører til flere kommersialiseringer. I snart to år har Inven2 jobbet målrettet med industrisamarbeid gjennom en rekke ulike prosjekter.
Les merSpydspiss innen bildediagnostikk
Oslo ImTECH har ambisjoner om å bli et av de ledende sentrene innen utvikling av bildediagnostikk og bildebehandling som brukes på sykehus verden over. Denne teknologien er avgjørende for å gi pasienter best mulig behandling.
Mer om Imtech
Kliniske studier er utprøving av nye medisiner og medisinsk utstyr for behandling på mennesker. Studier kan gjøres på både friske frivillige og pasienter. Kliniske studier gir helsepersonell viktig erfaring med nye legemidler og behandlingsmetodikk, forskningserfaring, samt internasjonalt nettverk.
Les mer
2018 har vært et svært innholdsrikt år for Inven2. Vi har hatt stor aktivitet og oppnådd mange av våre målsettinger for å ta forskningen til produkter eller tjenester på markedet.
Er det én ting jeg vil trekke fram som er spesielt gledelig så er det at mange av selskapene vi har etablert gjør det veldig bra i skarp global konkurranse. Vår portefølje av innovative bedrifter er ved årsskiftet verdt 10 milliarder kroner.
Les merVi gjør en forskjell ved å omgjøre forskning og kunnskap til samfunnsnyttige produkter og tjenester.

Forskning og kunnskap er nøkkelen til å løse globale samfunnsutfordringer innen helse, klima og miljø. Dyktige forskere og klinikere fra halve Norge melder inn idéer og oppdagelser til oss. Vi utvikler og forvalter idéene, og går videre med idéer vi kan omgjøre til samfunnsnyttige produkter og tjenester.
Vi har startet bedrifter som utvikler bedre kreftbehandling, bekjemper antibiotikaresistens og identifiserer sikkerhetsbrister i komplekse IT-systemer. To tredjedeler av våre bedrifter og lisenser er innen livsvitenskap, da vårt økosystem er spesielt sterkt innen dette fagfeltet.
En annen viktig oppgave vi har, er å forvalte avtaler om kliniske studier på vegne av sykehusene i vår region. Vi jobber for at flere nasjonale og internasjonale selskaper skal legge sine kliniske studier til Norge. Slik kan mer nyskapende behandling komme flere pasienter i Norge til gode.
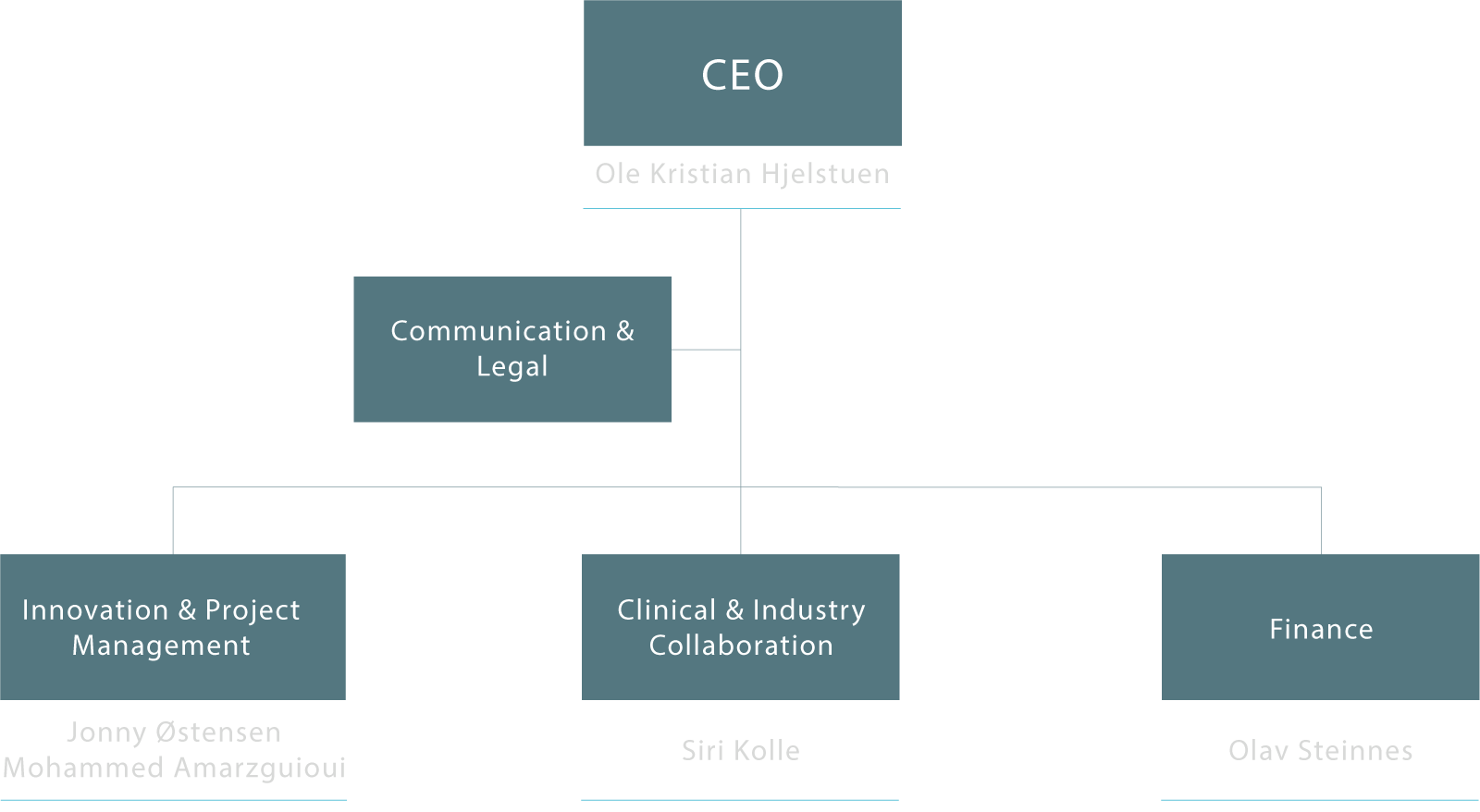
Inven2 er et aksjeselskap eid av Universitetet i Oslo og Oslo universitetssykehus. Selskapet har 34 ansatte.

Vi er stolt medarrangør av Cutting Edge-festivalen, Norges største vitenskaps- og teknologievent, hvor vi sammen med våre partnere Oslotech, Universitetet i Oslo og Conventor viser frem det siste innen vitenskap, teknologi og innovasjon.
www.cuttingedgefestival.no Se filmen fra Cutting EdgeBesøkende
Foredrag
Workshops
Debatter
1-1 møter
Utstillere

Nesten 90 1:1 møter mellom forskere, etablerte bedrifter og oppstartsbedrifter ble gjennomført under Cutting Edge i år. «Vi fikk viktige tilbakemeldinger,» forteller forsker Henriette Andresen etter møte med Pfizer.
Les merInven2 har en portefølje bestående av 45 selskaper og fem nye ble etablert i 2018. Samlet har selskapene en verdi på omtrent 10 milliarder norske kroner, og den øker stadig. Du kan lese mer i dybden om Vaccibody, Meshcrafts og Catalysts nedenfor.
Nordic Nanovector fikk ny administrerende direktør i 2018, Eduardo Bravo, med bred internasjonal erfaring. Selskapet er nå i gang med sin registreringsstudie av sin kreftbehandling av lymfekreft.
Går alt etter planen så vil de være på markedet med denne norskutviklete behandlingen i 2020. Et annet selskap som utvikler kreftbehandling er Ultimovacs. De kjøpte opp i 2018 teknologidelen av et svenskt selskap for over 50 millioner kroner og er nå godt i gang med studier av sin behandlende kreftvaksine i USA. Selskapet planlegger nå en børsnotering. Sist, men ikke minst, er selskapet Zelluna Immunotherapy. De utvikler kreftbehandling basert på en type immunterapi som kalles TCR, som handler om å sette inn såkalte super-T-celler inn i kreftpasienter slik at immunforsvaret selv blir i stand til å gjenkjenne og drepe kreftcellene.
Inven2 ønsker å gjøre en forskjell ved å omgjøre forskning og kunnskap til samfunnsnyttige produkter og tjenester. Siden oppstart har vi bidratt til at mange produkter og tjenester har kommet samfunnet til gode. Her er et utvalg av dem.
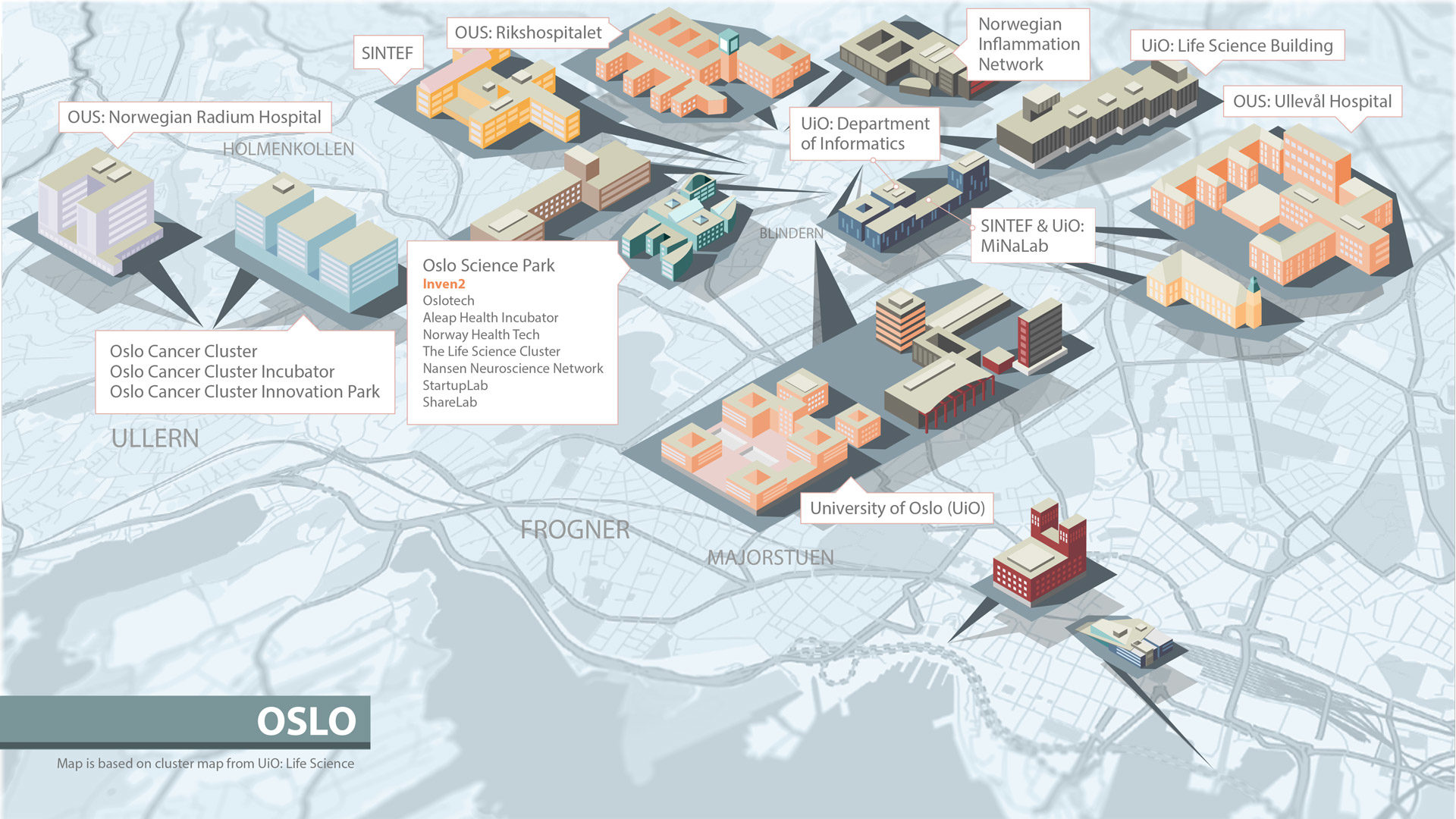
Inven2 er en del av et vitalt og stadig større økosystem innen forskning og utvikling. Bare i nærområdet til Inven2s lokaler i Forskningsparken finnes en rekke klynger og inkubatorer, samt Universitetet i Oslo og Oslo universitetssykehus.
Les mer